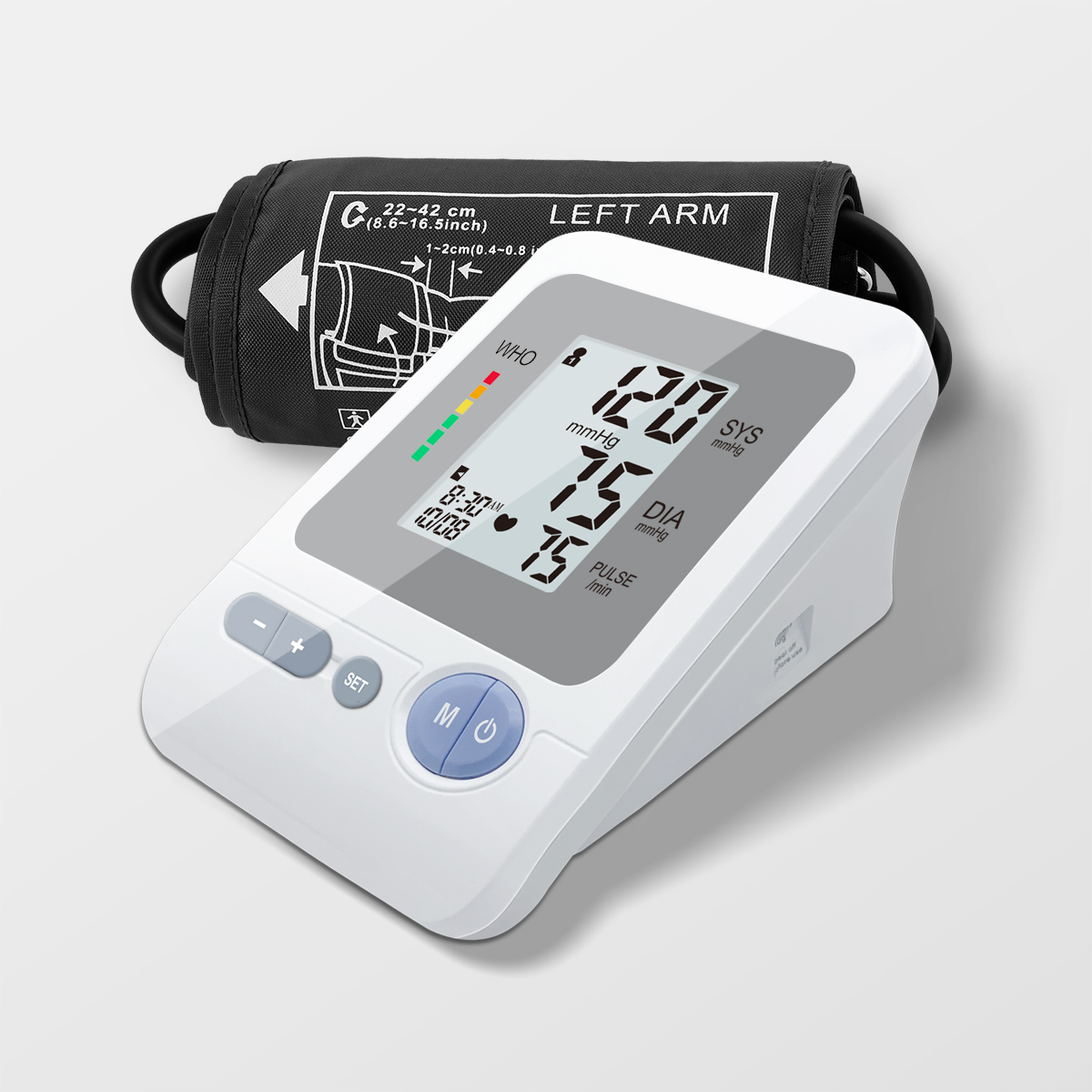
E-mail hmangin:
marketing@sejoy.com
Thupui pawimawh tak tak:
- Zavai
- Product Hming
- Product Keyword a ni
- Product Model hmanga siam a ni
- Product Summary chungchang
- Product Description chungchang
- Field hrang hranga zawn a ni
Please Choose Your Language
- English
- العربية
- Français
- Русский
- Español
- Português
- Deutsch
- italiano
- 日本語
- 한국어
- Nederlands
- Tiếng Việt
- ไทย
- Polski
- Türkçe
- አማርኛ
- ພາສາລາວ
- ភាសាខ្មែរ
- Bahasa Melayu
- ဗမာစာ
- தமிழ்
- Filipino
- Bahasa Indonesia
- magyar
- Română
- Čeština
- Монгол
- қазақ
- Српски
- हिन्दी
- فارسی
- Kiswahili
- Slovenčina
- Slovenščina
- Norsk
- Svenska
- українська
- Ελληνικά
- Suomi
- Հայերեն
- עברית
- Latine
- Dansk
- اردو
- Shqip
- বাংলা
- Hrvatski
- Afrikaans
- Gaeilge
- Eesti keel
- Māori
- සිංහල
- नेपाली
- Oʻzbekcha
- latviešu
- অসমীয়া
- Aymara
- Azərbaycan dili
- Bamanankan
- Euskara
- Беларуская мова
- भोजपुरी
- Bosanski
- Български
- Català
- Cebuano
- Corsu
- ދިވެހި
- डोग्रिड ने दी
- Esperanto
- Eʋegbe
- Frysk
- Galego
- ქართული
- guarani
- ગુજરાતી
- Kreyòl ayisyen
- Hausa
- ʻŌlelo Hawaiʻi
- Hmoob
- íslenska
- Igbo
- Ilocano
- Basa Jawa
- ಕನ್ನಡ
- Kinyarwanda
- गोंगेन हें नांव
- Krio we dɛn kɔl Krio
- Kurdî
- Kurdî
- Кыргызча
- Lingala
- Lietuvių
- Oluganda
- Lëtzebuergesch
- Македонски
- मैथिली
- Malagasy
- മലയാളം
- Malti
- मराठी
- ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
- Mizo tawng
- Chichewa
- ଓଡ଼ିଆ
- Afaan Oromoo
- پښتو
- ਪੰਜਾਬੀ
- Runasimi
- Gagana Samoa
- संस्कृत
- Gaelo Albannach
- Sepeti
- Sesotho
- chiShona
- سنڌي
- Soomaali
- Basa Sunda
- Wikang Tagalog
- Тоҷикӣ
- Татарча
- తెలుగు
- ትግንያውያን
- Xitsonga
- Türkmençe
- संस्कृत
- ئۇيغۇرچە
- Cymraeg
- isiXhosa
- ייִדיש
- Yorùbá
- isiZulu